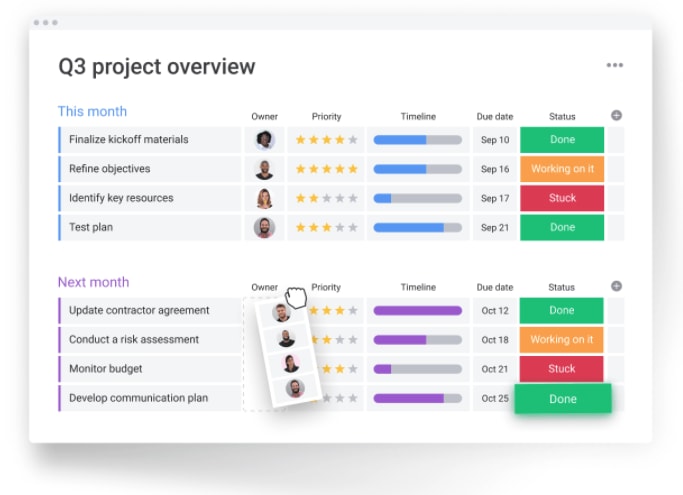
 Manufacturers are often subject to an alphabet soup of regulatory standards (FDA, ISO 9001, Title 21 CFR Part 11, NERC, SSOP, HAACP, and more). Strong QMS is assisting manufacturers in meeting some of their most critical business challenges: Ensuring Regulatory Compliance Reporting and Business Intelligence: Reference key performance indicators and performance dashboards to turn quality management data into actionable business intelligence.Ī corrective actions chart in TRACKMEDIUM.Document Management: Coordinate the data management challenge, facilitate collaboration, and ensure easy access to important quality management documentation.Audits and Inspections: Define, schedule, and execute audits and inspections.Customer Complaint Management: Assist service personnel in quickly resolving customer issues.Ensure consistent process reviews are happening Approval Management: Submit requests and manage approvals.May include more sophisticated monitoring controls that integrate with operational equipment. Track a database of standardized parts metrics. Parts Non-conformance: Define the dimension your parts are measured upon for non-conformance. 
Corrective Action/Preventative Action (CAPA): Includes task assignments, reporting, and monitoring to ensure that the right response has been implemented when quality has deviated outside of pre-identified standards.Workflow Management: Define all processes involved in manufacturing an end-product. 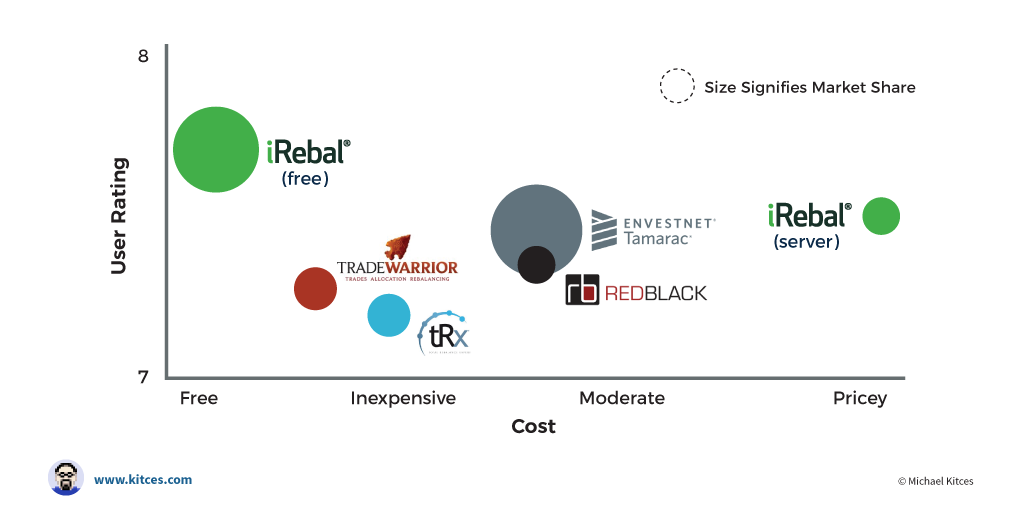
Predict failure and service rates, as well as their financial implications. Risk Management and Analysis: Create what-if scenarios to reliably analyze potential costs related to quality exceptions.Quality Planning: Set quality objectives across a variety of dimensions including cycle times, scrap/waste percentages, defect rates, measurement deviations, durability metrics, and more.Compliance Control: Includes documentation, planning, scheduling, reporting, and auditing related to adhering to regulatory standards.Core functions and features often include: Quality management system (QMS) software offers a wide variety of functionality. Features of Quality Management Software (QMS) (See: What is EQMS?) Key indicators on the uniPoint dashboard. EQMS assists businesses with globalization by enhancing their quality management practices across the supply chain. These options may be general-use for all manufacturers, or focus on quality management capabilities unique to specific manufacturing industries (such as pharmaceuticals or chemicals).Įnterprise-level quality management systems (EQMS) have a greater scope than generic QMS solutions. QMS software is used on conjunction with other manufacturing software or MRP software options. 
An integrated approach that allows data to flow between each area is required. They’re a structural impediment to achieving your quality goals. Distributed systems are inherently inefficient. It’s no longer enough to have engineering, the production floor, purchasing, sales, and service working independently to improve quality. Quality management software (QMS) offers a comprehensive tool kit to analyze risks, set quality objectives, implement workflows to achieve standards, and audit for optimal performance.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
- Blog
- New Page
- Home
- Sample Debut Party Program Script
- MANCITY ANTHEN MP3
- Bioestadistica Daniel 4 Edicion Pdf: Software Free Download
- Download Internet Speed Meter Pro Untuk Android
- Download Game Digimon World For Android
- Unity3d Racing Game Source
- Download Original Soundtrack Naruto The Movie Rar
- Pro Cycling Manager 2011 Completo Pc
- Tamil Hd Video Songs Free Download
- Best Ip Booter Download Free
- Becker Traffic Assist Highspeed Ii Kartenupdate
- How To Crack Idm Using Ollydbg
- Sublime 1996 Rar
- Sims 2 Stealth Hoodsdownload Free Software Programs Online
- Canon Ir C2880 C3380 Pcl6 Drivers For Mac
- Rafoo Chakkar Full Movie 1975 Hd Download
- Hillsong Let It Rain Mp3 Download
- God Bless Amerika Lil Wayne Download Sharebeast
- Anbulla Snegithi Sun Tv Serial
- Confirmation Id Visio 2010
- Download Doa Pengobral Dosa
- Multicast Traffic Generator Windows Movie
 RSS Feed
RSS Feed
